From University of Illinois Chicago (UIC) over 80 bio-friendly compositions with superior freeze-protection properties

by Rukmava Chatterjee – University of Illinois Chicago
The sight of snowflakes falling in thousands from the winter sky is a delightful experience until its dreary aftermath. The buildup of ice and frost on functional surfaces and its stubborn adherence pose a huge problem for a wide spectrum of industries ranging from energy to transportation systems.
Energy research reports in the past decade have estimated that severe ice storms account for electric power outages as high as 10%, impacting the lives of 3 million people in the United States every winter. In general, the undesired consequences of icing/frosting has an irrefutable effect on quotidian human activities.
Naturally, technologies that can delay ice/frost formation on surfaces or facilitate its passive removal play a pivotal role in industrial applications operating in frigid climates. Consequently, the financial market for anti-icing & icephobic materials amounts to 20 billion USD annually.
Passive freeze-protection techniques inefficient, expensive and environmentally unfriendly
Unfortunately, most of the passive ice-shedding techniques typically employed, either bear expensive fabrication costs or suffer from poor long-term durability. Because of these inherent challenges, they have not seen widespread commercial adoption. Hence, active techniques like electrothermal heating, chemical de-icing, and good-old mechanical scraping remain the industry-wide foundation to mitigate icing/frosting problems despite being energetically expensive and having a negative environmental footprint.
Amongst these, anti/de-icing chemicals particularly monopolize the billion-dollar market share because of their easy availability and quick applicability. For example, dowsing airplane wings with thousands of gallons of anti-freeze glycols to prevent ground icing and ensure their safe take-off is a common sight during the chilly winters.
But being liquid, these chemicals are easily blown away or dissolved thereby making them temporary measures.
Washed away, they end up in the local freshwater streams polluting them and disturbing the marine ecological balance.
The solution to the problem
To address these burning issues, University of Illinois Chicago (UIC) researchers have formulated an extensive library of more than eighty compositions with superior freeze-protection properties.
The library of formulations can be broadly classified as polymeric solutions, emulsions, creams, and gels. The skeleton of the developed coatings is comprised of dimethyl sulfoxide, a naturally derived, bio-friendly cryoprotectant possessing miraculous medicinal properties that was securely locked in a polymeric matrix, which grants it enhanced resistance against dissolution and shear-driven removal. Compared to untreated industrial metal surfaces, these novel coatings are able to simultaneously delay ice formation (~87 times longer) while ensuring facile ice shedding (~665 times easily); a complete game-changer when compared to most of the commercial coatings available in today’s market (Figure 1).
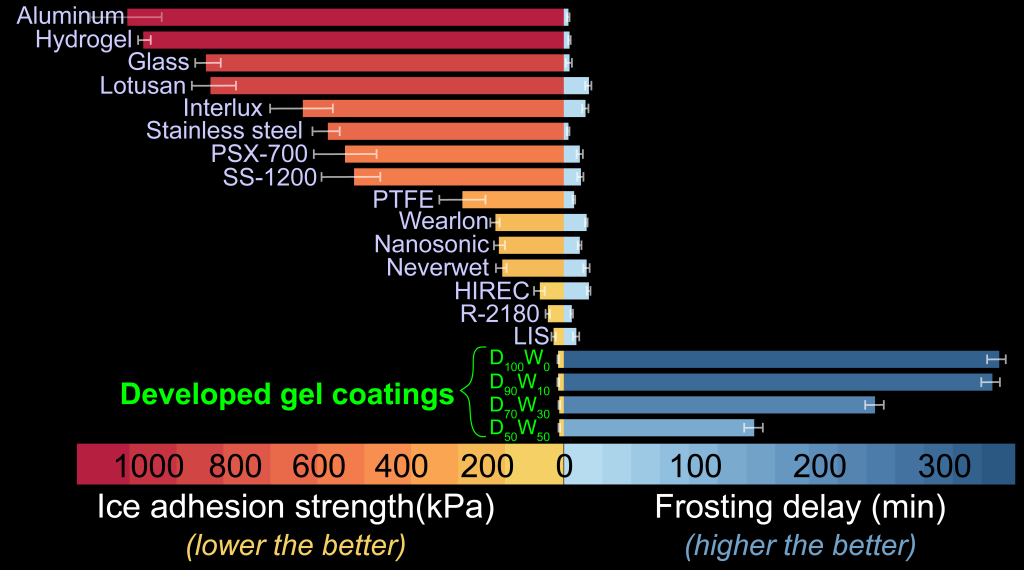
As the world gradually transitions towards sustainable & renewable sources of energy, it is critical to equip the involved power-horses (e.g., wind turbine, solar photovoltaics, power transmission lines) with appropriate winter-weather packages for fail-safe performance in cold ambientes.
In the colder states of America, Canada, and Europe, where winter temperature plummets well below freezing, icing on wind turbine blades causes them to harness power less efficiently.
The winter gear typically employed to prevent icing includes electrothermal heating. Ironically enough, this implies consuming energy to generate energy. In their latest study, the UIC researchers demonstrate the efficacy of their developed coatings for various applications ranging from wind turbine/airplane propellers to power transmission cables.
In their experiment (Figure 2), the scientists subjected two scaled-down propeller blades, one bare (left) and the other coated to simulated winter weather conditions. A few minutes into the experiment, the bare propeller blade was seen to turn white from the ice accretion and ‘ice-throwing’ incident was observed.
The term ‘ice throw’ is a euphemism in the wind energy industry for the phenomenon whereby chunks of ice are ejected off the rotating propellers. With gradual frost formation on the propellers, a time comes when its load-bearing threshold is compromised, resulting in the accumulated ice shards to be shed by gravitational and centrifugal forces. These dislodged frozen scrapings falling from the sky are injurious to neighboring human life, automobiles, and infrastructures.

Image credit: Rukmava Chatterjee
In our experiments, analogous behavior was observed for the bare propeller. Surface frosting and ice-shedding commenced in the first 6 minutes of the test on the uncoated blades, with the heavily frosted blades coming to a halt after ~28 minutes of constant operation.
Maintaining the transparency of the surfaces
Functional performance aside, a high degree of optical transparency is one of the fundamental requirements of various optical applications like architectural windows, solar panels/photovoltaic devices, automotive windshields, painted structures, and optoelectronic devices.
However, exposure to environmental precipitation such as ice, frost, wet snow, etc., fouls the aforementioned surfaces impairing their transparency, and vitiates their basic functionality. However, optical transparency and anti-icing are two competitive properties that need integration for such applications’ operability in inclement weather conditions. The majority of the superhydrophobic surfaces intended for anti-icing rely on intricate micro/nano-surface texturing. However, microstructured surfaces possessing surface features larger than the visible wavelength of light are not transparent and look opaque due to Mie scattering from their rough textures.
An elementary requirement for a transparent coating is that the constituents of the same should be made up of low-light-absorbing materials with an intermediate refractive index between that of air and the base substrate (for example, glass).
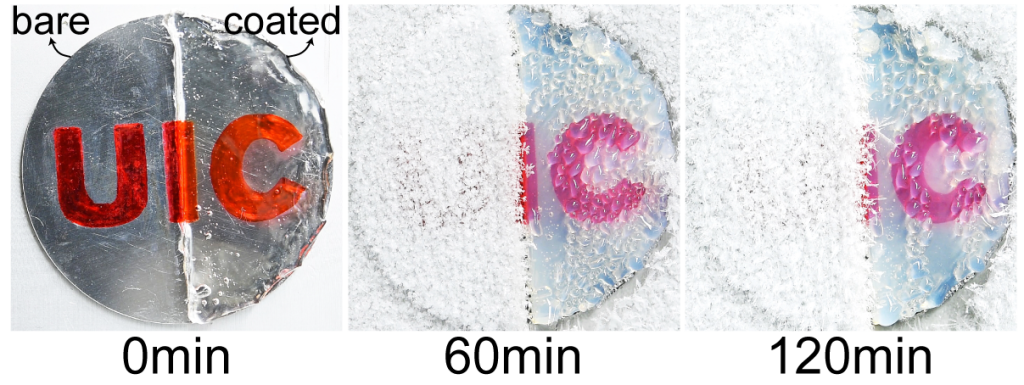
Steering clear of the use of any micro-texturing and considering other constraints, the UIC developed anti-freezing gel coating renders high optical transparency under heavy icing/frosting conditions. The researchers verified this by coating different industrial materials (glass, aluminum, steel, Teflon, etc.) with their coatings of varying thickness and conducting inspections of transparency during icing tests. Figure 3 shows a demonstrative example whereby after 60 minutes of continued frosting, the uncoated portion of an aluminum disc was covered in a thick sheet of frost, making the red UIC logo indiscernible. However, even after 2 hours, the gel-coated half of the disc provided an unobstructed view of the underlying logo.
The coatings are multifunctional by design. They function by leveraging certain thermoresponsive properties, allowing them to generate a slippery lubricating layer on their surface which acts as a protective barrier against contaminants ranging from minuscule bacteria to macroscale frost. The implications of this are huge. For example, the same coating that can prevent a smartwatch’s battery from dying in the frigid winter can concomitantly preclude any bacterial contamination. Due to the versatile commercial applications, a worldwide patent application titled “Compositions and Methods for Inhibiting Ice Formation on Surfaces” has been filed by UIC’s Office of Technology Management.
Further information can be found in the original research article: A Family of Frost-Resistant and Icephobic Coatings, Rukmava Chatterjee, Hassan Bararnia, Sushant Anand.
DOI: https://doi.org/10.1002/adma.202109930
Written by Rukmava Chatterjee, Department of Mechanical & Industrial Engineering, University of Illinois at Chicago – for any question





